FDA Disclaimer: Thymosin Alpha-1 is not approved by the FDA for bodybuilding, anti-aging, or general wellness purposes. It is available only as a research chemical. The pharmaceutical form (Zadaxin/thymalfasin) is approved in some countries for specific medical conditions but not in the United States for most indications. This article is for informational purposes only and does not constitute medical advice.
Thymosin Alpha-1 Benefits: What the Research Shows About This Immune Peptide
TL;DR: Thymosin Alpha-1 (Ta1) is a 28-amino-acid peptide produced naturally in the thymus gland and studied for immune signaling. The literature includes hepatitis, oncology-adjunct, and immune-compromised research settings, but US readers should treat that evidence as clinical-context data, not a blank check for self-experimentation or treatment claims.
In the US, Ta1 is sold only as a research chemical, and the published safety literature is not the same thing as FDA approval.
Table of Contents
- What Is Thymosin Alpha-1?
- How Thymosin Alpha-1 Works: Mechanism of Action
- Immune System Benefits
- Research in Hepatitis B and C
- Cancer Adjunct Therapy Research
- Aging and Immune Function
- Other Exploratory Research Areas
- Clinical Study Ranges Reported in the Literature
- Safety and Side Effects
- Legal Status
- Frequently Asked Questions
What Is Thymosin Alpha-1?
Understanding thymosin alpha-1 benefits starts with what it is: a small peptide - 28 amino acids in length - produced naturally by the thymus gland. The thymus sits behind the sternum and plays a central role in training the immune system during early life. Ta1 was first isolated in the 1970s by researchers studying thymic hormones and their effects on immune cell development.
A synthetic version called thymalfasin (marketed as Zadaxin) has been used clinically in some countries within narrow hepatitis and adjunct-care settings. But in the United States, it remains unapproved for most indications and is sold strictly as a research peptide.
The peptide belongs to a broader family of thymosins - proteins secreted by the thymus that help regulate immune cell production and function. Unlike some other peptides in the research space, Ta1 has a substantial body of published clinical literature behind it, including randomized controlled trials.
If you are comparing research peptide vendors, check our best peptide companies guide for vendor reviews and quality benchmarks.
Research-Grade Thymosin Alpha-1 Available Now
60+ third-party tested research peptides including Thymosin Alpha-1
60+ third-party tested research peptides and stacks
How Thymosin Alpha-1 Works: Mechanism of Action
Ta1 does not work like a typical drug that blocks a receptor or inhibits an enzyme. Instead, it acts as an immunomodulator - it helps coordinate and fine-tune the immune response through several pathways.
Toll-Like Receptor Activation
The primary mechanism involves activation of Toll-like receptors (TLRs). Ta1 functions as an agonist for TLR2, TLR3, TLR4, TLR7, and TLR9 on both myeloid and plasmacytoid dendritic cells. This TLR engagement triggers downstream signaling through the p38 MAPK and NF-kB pathways, which are fundamental to immune cell activation and cytokine production (Romani et al., Current Pharmaceutical Design, 2004; Li et al., Expert Opinion on Biological Therapy, 2005).
T-Cell Stimulation
Ta1 promotes the differentiation and maturation of T-cell progenitor cells in the thymus. It increases CD4+ helper T cells, CD8+ cytotoxic T cells, and improves the CD4+/CD8+ ratio - a key marker of immune health. It also stimulates IL-2 receptor expression on T cells, which drives T-cell proliferation and activation (King & Tuthill, Clinical Immunology, 2016).
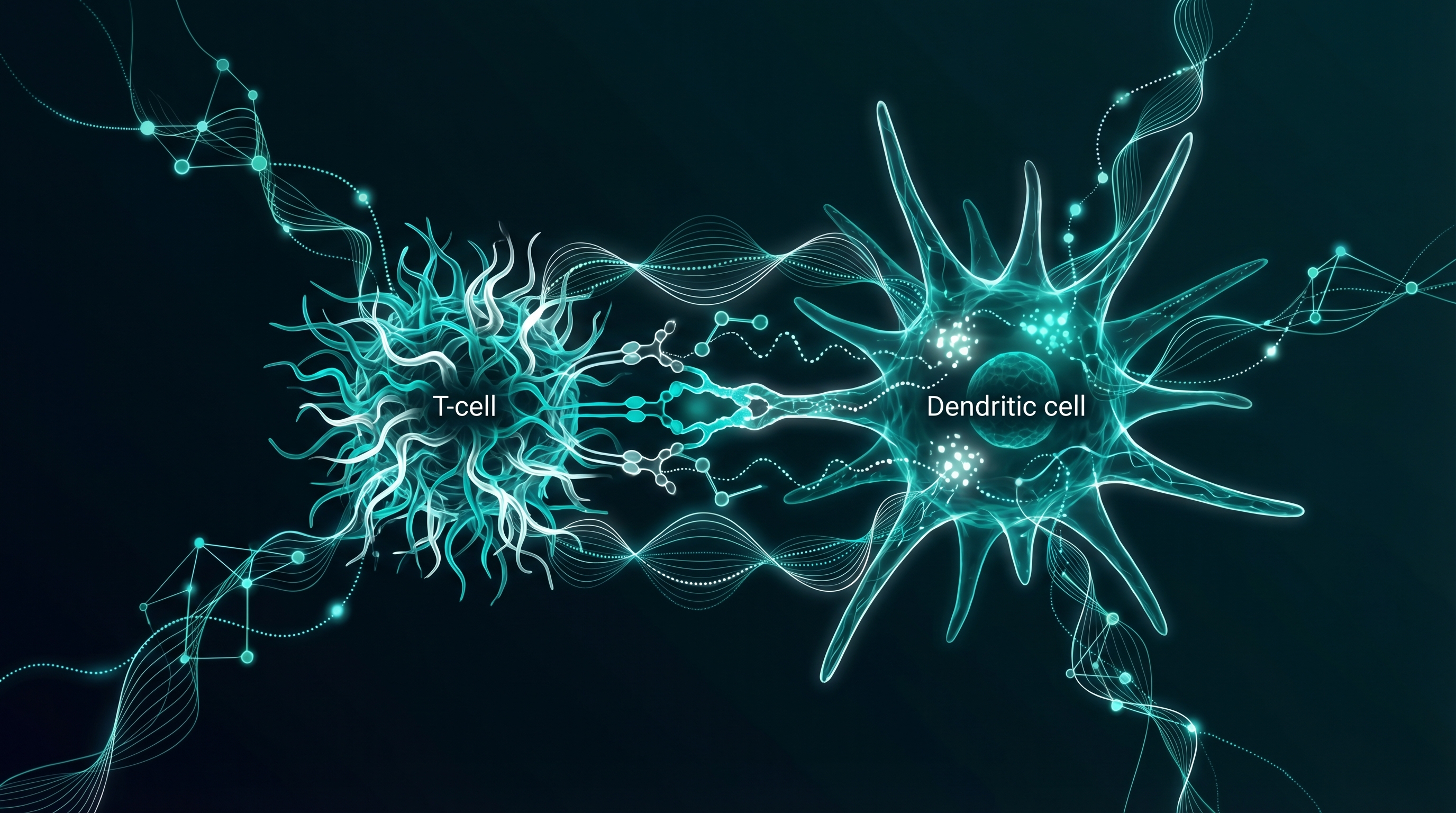
Dendritic Cell Maturation
Dendritic cells act as sentinels. They patrol the body, capture antigens, and present them to T cells to trigger adaptive immunity. Ta1 upregulates surface markers on immature dendritic cells (CD40, CD80, MHC class I and II), pushing them toward full maturation. Mature dendritic cells treated with Ta1 show significantly improved ability to stimulate T-cell proliferation (Li et al., 2005; PMC1986709).
Natural Killer Cell Enhancement
Natural killer (NK) cells identify and eliminate virus-infected cells and early cancer cells without needing prior antigen exposure. Research suggests Ta1 enhances NK cell cytotoxicity, adding another layer to its antiviral and antitumor effects.
For help calculating dosages for research applications, use our free peptide reconstitution calculator.
Immune System Benefits
The immune-related benefits of Thymosin Alpha-1 are broad because the peptide targets multiple components of the immune system simultaneously. Rather than suppressing one pathway (like corticosteroids do), Ta1 helps restore balance.
Research published in Expert Opinion on Biological Therapy and reviewed in a 2020 Clinical Therapeutics comprehensive review (PMC7747025) identified these primary immune effects:
- T-cell restoration: Increases CD3+, CD4+, and CD8+ T lymphocyte counts, particularly relevant in immunocompromised states
- Improved vaccine response: Studies in elderly populations showed enhanced antibody production after influenza vaccination when Ta1 was co-administered
- Cytokine balance: Promotes production of IL-2, IL-12, and interferon-gamma (Th1-type) while also supporting IL-10 (regulatory), creating a balanced immune response
- Reduced inflammatory markers: A 2025 meta-analysis in Frontiers in Immunology found Ta1 significantly reduced inflammatory markers in severe acute pancreatitis patients
- Antimicrobial defense: Enhances the body's ability to fight viral, bacterial, and fungal infections through coordinated innate and adaptive immune activation
One detail that researchers find interesting is the peptide's dual nature. Ta1 can amplify immune responses when they are weak (helping fight infections) but also modulate overactive inflammation. This bidirectional effect is what makes it distinct from simple immune stimulants.
Research in Hepatitis B and C
The most extensive clinical research on Thymosin Alpha-1 involves viral hepatitis, particularly hepatitis B (HBV) and hepatitis C (HCV).
Hepatitis B
A Phase III multicenter, randomized, double-blind, placebo-controlled study tested Ta1 as monotherapy in chronic HBV patients. Subjects received 1.6 mg subcutaneously twice weekly for 26 weeks. The complete response rate was 14% for Ta1 versus 4% for placebo (Mutchnick et al., Hepatology, 1999). While statistically significant, the modest absolute improvement led researchers to focus more on combination approaches.
Later studies explored Ta1 combined with interferon-alpha and nucleoside analogs. Results improved meaningfully: one study reported HBV DNA clearance rates of approximately 40.6% at 26 weeks and 26.5% at 52 weeks with combination therapy (PMC7747025).
Hepatitis C
As monotherapy, Ta1 showed limited efficacy against HCV. But in combination with interferon-alpha 2b, results were stronger. One trial reported that 71% of patients achieved normal ALT levels and 65% achieved HCV RNA clearance with the combination, compared to interferon alone (Sherman et al., Journal of Viral Hepatitis, 2007).
Ta1 is currently in Phase III trials for hepatitis C. The synthetic form (thymalfasin/Zadaxin) is approved for hepatitis B treatment in several Asian and South American countries.
Cancer Adjunct Therapy Research
Ta1 has appeared in adjunct-care oncology literature, meaning it was studied alongside conventional treatments like chemotherapy or radiation in tightly controlled clinical contexts. That is not the same as evidence that it treats cancer on its own, and readers should be careful not to flatten exploratory adjunct data into a general-use claim.
Non-Small Cell Lung Cancer (NSCLC)
A 2022 study published in Translational Lung Cancer Research reported better lymphocyte recovery and lower pneumonitis rates in one NSCLC adjunct-care setting. Useful signal, yes. But this is still specialty oncology literature, not a consumer-facing treatment takeaway.
Hepatocellular Carcinoma (HCC)
After liver resection for HCC, one study associated Ta1 use with stronger survival metrics and recurrence-free survival. That kind of signal is worth noting, but it still sits inside post-surgical oncology care rather than general wellness use (Shu et al., 2022).
Melanoma and Breast Cancer
A 2018 study in Scientific Reports (Nature) tested a modified Ta1-Fc fusion protein with extended half-life. The construct showed immune-modulating activity and slowed progression of both melanoma and breast cancer in animal models. While preclinical, the results support further investigation of Ta1-based approaches in oncology.
Advanced/Refractory Solid Tumors
One small advanced-solid-tumor study reported measurable disease-control signals, but this is the kind of early or adjunct evidence that needs restraint in how it is presented. It belongs in a research summary, not in a promise-heavy sales frame (PMC7747025).
Access 118+ Research Peptides in One Place
Including Thymosin Alpha-1 in injectable, spray, and capsule forms
Create Your Free Limitless Account →
118+ research peptides in injectable, spray, and capsule forms - create a free account to access the full catalog
Use code PeptidePick for a discount at checkout
Aging and Immune Function
As people age, the thymus gland progressively shrinks - a process called thymic involution. By age 60, the thymus produces only a fraction of the T cells it did in youth. This decline contributes to weaker immune responses, poorer vaccine efficacy, and higher susceptibility to infections and cancer in older adults.
A 2025 review in the International Journal of Molecular Sciences examined Ta1's potential to counteract age-related immune decline. The findings suggest that Ta1 can:
- Stimulate thymic output of new T cells, partially compensating for thymic involution
- Improve vaccine response in elderly subjects by enhancing dendritic cell-T cell interactions
- Restore CD4+/CD8+ ratios that decline with age
- Reduce inflammatory markers associated with "inflammaging" - the chronic low-grade inflammation common in aging
But this research is still early. Most aging-related Ta1 studies are small or preclinical. The data is promising enough to warrant larger trials, but the evidence is not yet strong enough to make firm claims about longevity or anti-aging effects.
For related reading on longevity peptides, see our best peptides for anti-aging guide and our Epithalon benefits article.
Other Exploratory Research Areas
COVID-19 and Severe Infections
During the COVID-19 pandemic, researchers investigated Ta1 as a potential treatment for severely ill patients. The rationale was that Ta1 could help repair lymphopenia (dangerously low lymphocyte counts) and calm the cytokine storm seen in severe COVID cases. Clinical trials were registered in China, and early data suggested improvements in lymphocyte recovery. A full peer-reviewed analysis is still pending for many of these studies.
Sepsis
Ta1 has also appeared in sepsis literature, where researchers are looking at whether immune modulation can improve outcomes in highly acute settings. That remains a specialized hospital-level question, not a broad consumer use case.
Lyme Disease
Some integrative practitioners have used Ta1 off-label as part of Lyme disease protocols, aiming to boost immune recognition of Borrelia burgdorferi. The published evidence for this application is thin - mostly case reports and theoretical rationale based on Ta1's general immune-enhancing properties.
Severe Acute Pancreatitis
A 2025 systematic review and meta-analysis in Frontiers in Immunology pooled severe acute pancreatitis studies and reported lower inflammatory markers plus lower infection rates. Even so, this remains condition-specific hospital literature and should be read with that scope in mind.

Clinical Study Ranges Reported in the Literature
This section is here to show how Ta1 appeared in published clinical literature, not to provide a self-use protocol. The reported ranges vary by study context, patient population, and local regulatory setting.
| Condition | Dose | Frequency | Duration |
|---|---|---|---|
| Chronic Hepatitis B | 1.6 mg | Twice weekly (SC) | 26-52 weeks |
| Chronic Hepatitis C (combo) | 1.6 mg | Twice weekly (SC) | 24-48 weeks |
| Cancer adjunct (NSCLC) | 1.6 mg | Twice weekly (SC) | During chemo/radiation |
| Immune support (elderly) | 0.8-1.6 mg | Twice weekly (SC) | Variable |
These ranges come from published research and are included only to show how the literature was structured. They are not self-administration recommendations, and they should not be treated like a consumer protocol.
Safety and Side Effects
Published review literature often describes Ta1 as generally well tolerated in the specific clinical settings where it has been studied. That is useful context, but it is not the same thing as a broad FDA-approved safety profile for general use.
Common Side Effects
- Mild injection site reactions (redness, discomfort, swelling)
- Fatigue
- Headache
- Mild gastrointestinal discomfort
Less Common
- ALT flares in hepatitis patients (usually transient, rarely requiring treatment discontinuation)
- TSH abnormalities in hepatitis C patients
- Rare reports of fever, muscle aches, or nausea when combined with interferon
Ta1 is contraindicated in patients undergoing deliberate immunosuppression (such as organ transplant recipients on anti-rejection drugs) and should be used cautiously in pregnant or nursing women due to insufficient safety data.
The fact that it has been administered to thousands of patients across multiple countries with a consistently mild side-effect profile is meaningful. But it is not risk-free, and the long-term effects of chronic administration outside clinical settings have not been well-studied.
For broader peptide safety information, see our peptide side effects overview.
Legal Status
In the United States, Ta1 is sold as a research chemical only. The FDA has not approved it for general medical use. The pharmaceutical-grade form (thymalfasin/Zadaxin) by SciClone Pharmaceuticals has Orphan Drug designation for some cancer indications but is not widely approved in the US.
In contrast, Ta1 is approved for clinical use in over 35 countries, primarily in Asia and South America, where it is used for hepatitis B, as a cancer adjunct, and for immune support in immunocompromised patients.
For US researchers purchasing Ta1, it falls under the same regulatory framework as other research peptides. See our are peptides legal guide for more detail on peptide regulations.
Third-Party Tested Thymosin Alpha-1
99% pure research peptides with independent lab verification
99% pure research peptides - use code Peptidepick15 for 15% off
Use code Peptidepick15 for 15% off your order
For readers interested in oral supplement alternatives that support immune function without injections, Nootropics Depot offers third-party tested supplements like medicinal mushroom extracts, elderberry, and vitamin D that complement general immune health protocols.
Frequently Asked Questions
Is Thymosin Alpha-1 the same as Thymosin Beta-4?
No. Despite the similar names, these are entirely different peptides with different functions. Thymosin Alpha-1 primarily modulates immune function through T-cell and dendritic cell activation. Thymosin Beta-4 (and its synthetic fragment TB-500) is involved in tissue repair, wound healing, and cell migration.
They work through completely different mechanisms.
See our TB-500 guide and BPC-157 vs TB-500 comparison for more on the tissue-repair side.
How long does Thymosin Alpha-1 take to work?
In clinical studies, measurable immune changes (increased T-cell counts, improved CD4+/CD8+ ratios) were typically observed within 2-4 weeks of twice-weekly administration. However, clinical outcomes in conditions like hepatitis often required 6 months or longer to evaluate. The timeline depends heavily on what is being measured and the patient's baseline immune status.
Can Thymosin Alpha-1 be used preventively?
This is a gray area. Some anti-aging and integrative medicine practitioners use Ta1 prophylactically in older adults to counteract immune senescence. The published research supports its ability to enhance vaccine responses and improve immune markers in elderly subjects.
But using it preventively in healthy younger adults lacks clinical evidence. The cost-benefit analysis is unclear for that population.
Does Thymosin Alpha-1 suppress the immune system?
No. Ta1 is an immunomodulator, not an immunosuppressant. It works by enhancing and coordinating immune responses, not by suppressing them. That said, its ability to promote regulatory T cells and IL-10 production means it can dampen excessive inflammation - but this is regulation, not suppression.
Is Thymosin Alpha-1 approved by the FDA?
Ta1 does not have full FDA approval for general use in the United States. The synthetic form (thymalfasin) has received Orphan Drug designation for certain cancer indications, which provides incentives for development but is not the same as approval. It is approved for clinical use in over 35 other countries.
What is the difference between Ta1 and thymalfasin?
Thymalfasin is the generic name for the synthetic, pharmaceutically manufactured version of Thymosin Alpha-1. They have the same amino acid sequence. The brand name for thymalfasin is Zadaxin, manufactured by SciClone Pharmaceuticals. When people refer to "Thymosin Alpha-1" in the research peptide market, they are referring to synthetically produced Ta1 that is sold for laboratory research, not the pharmaceutical-grade drug.
Can Thymosin Alpha-1 help with autoimmune conditions?
The evidence here is mixed and complicated. Because Ta1 stimulates immune activity, there is a theoretical concern that it could worsen autoimmune conditions. But its ability to promote regulatory T cells (which suppress autoimmune responses) creates a counterargument.
A 2016 study in the Journal of Cellular and Molecular Medicine (PMC5011367) examined serum Ta1 levels in autoimmune disease patients, but the clinical implications remain unclear. Anyone with an autoimmune condition should approach Ta1 with caution and medical supervision.
Related Articles
- Best Peptide Companies: Trusted Vendor Reviews
- Epithalon Peptide Benefits: Longevity Research Guide
- Peptide Side Effects: What Researchers Need to Know
- Are Peptides Legal? A State-by-State Guide
- What Are Peptides? A Complete Beginner Guide
- Peptide Therapy Cost: What to Expect
Affiliate Disclosure: PeptidePick earns commissions from qualifying purchases through affiliate links on this page. This does not affect our editorial content or product recommendations. All opinions are our own. Learn more in our full affiliate disclosure.